The SPORIFY Philosophy: Compliance Made Easy, Not Overengineered
Supporting the Integrity of
Global Regulatory Data
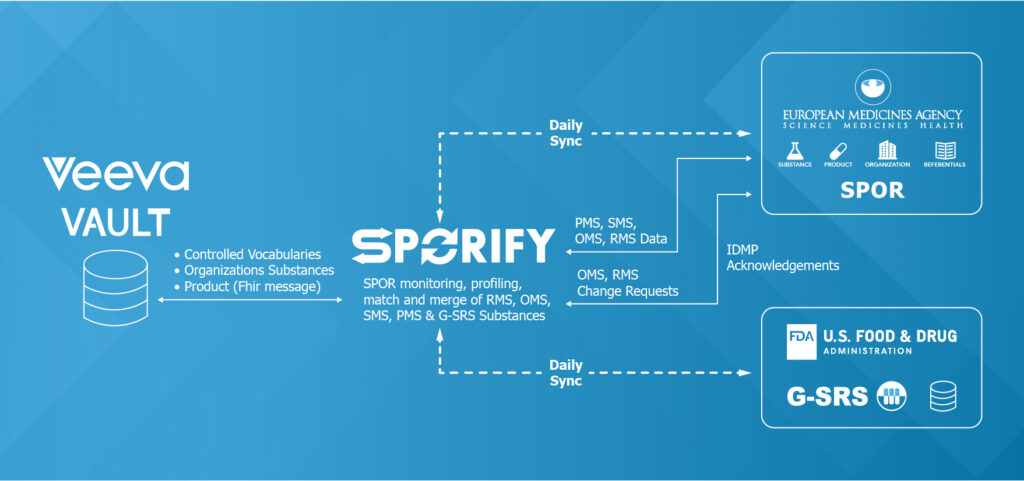
The mandate is clear: ISO IDMP standardization is no longer optional. However, the industry’s response has been split. Some vendors believe that the solution requires pharma companies to engage in extensive, multi-year projects to re-architect their entire existing data infrastructure.
It doesn’t have to be this way.
Since 2018, SPORIFY has championed the clearest path. Our solution is tailored to your operational realities, serving as a highly tactical, low-friction bridge that ensures your data is managed, compliant, and accepted globally – all without having to overhaul your systems.
SPORIFY is the pioneering and most mature IDMP solution on the market, trusted by over 25 customers globally, as well as the National Competent Authorities. Additionally, we are actively involved in the EMA SPOR Key User Group, UNICOM, EU-SRS project, SPOR Task Force, and actively participate with EMA on new development UAT to ensure our software aligns perfectly with the regulators’ actual expectations.
You need speed, simplicity, and consistency – and SPORIFY is purpose-built to meet those needs.
